1813 Telmisartan Tablets IP 80mg
Powerful Antihypertensive Solution
₹24.20
Telmisartan Tablets IP 80mg are designed to effectively control high blood pressure and support long-term cardiovascular well-being. By relaxing blood vessels, these tablets facilitate smoother blood flow and help reduce the risk of heart-related complications.
Description
Telmisartan is an angiotensin receptor blocker (ARB), It is used in the treatment of hypertension also effective in cardiovascular risk reduction.Telmisartan blocks the action of angiotensin II (Ang II), the primary effector molecule of the renin-angiotensin-aldosterone system (RAAS). It is the sixth of this class of 《sartans》 to be marketed after the lead compound Losartan. Its long lasting effect (24h half-life) could be the main difference with other angiotensin II antagonists. Unlike several other agents in this category, its activity does not depend upon transformation into an active metabolite, the 1-O-acylglucuronide being the principal metabolite found in humans. Telmisartan is a potent competitive antagonist of AT1 receptors that mediate most of the important effects of angiotensin II while lacking affinity for the AT2 subtypes or other receptors involved in cardiovascular regulation.
Chemical Properties
White or off white crystalline powder
Originator
Boehringer Ingelheim (Germany)
Uses
Telmisartan, an angiotensin II receptor antagonist, is an effective medication for the treatment of hypertension. It can be used alone or in combination with other antihypertensive drugs. Additionally, it is beneficial in the treatment of diabetic nephropathy in hypertensive individuals with type 2 diabetes mellitus. Telmisartan is also used to address congestive heart issues.
Definition
ChEBI: Telmisartan is a member of the class of benzimidazoles used widely in the treatment of hypertension. It has a role as an antihypertensive agent, an angiotensin receptor antagonist, an EC 3.4.15.1 (peptidyl-dipeptidase A) inhibitor, a xenobiotic and an environmental contaminant. It is a member of biphenyls, a member of benzimidazoles and a carboxybiphenyl.
Manufacturing Process
Telmisartan was synthesized through the reaction of methyl 3,4-diaminobenzoate dihydrochloride and butyric acid chloride in the presence of phosphorous oxychloride, with subsequent purification steps:
A solution of 23.9 g (100 mMol) of methyl 3,4-diaminobenzoate dihydrochloride and 11.7 g (110 mMol) of butyric acid chloride in 100 ml of phosphorus oxychloride is refluxed for 2 h. Then about 80 ml of phosphorus oxychloride are distilled off and the residue is mixed with about 150 ml of water. The oily crude product precipitated is extracted three times with 50 ml of ethyl acetate and after evaporation purified by column chromatography (600 g of silica gel; eluant:methylene chloride/methanol (30:1)). Yield of methyl-2-n-propyl-benzimidazole-5-carboxylate: 15.0 g of oil (69%).
A solution of 15.0 g (73 mmol) of methyl 2-n-propyl-benzimidazole-5- carboxylate and 8 g (200 mMol) of sodium hydroxide in 200 ml of water and 400 ml of ethanol is refluxed for 2 h. Then the alcohol is distilled off, the aqueous solution is acidified with dilute sulphuric acid (pH 4-5) and evaporated using a rotary evaporator. The product crystallising out is suction filtered, washed with 50 ml of acetone and 50 ml of diethylether and dried. Yield of 2-n-propyl-benzimidazole-5-carboxylic acid-hemisulphate: 9.1 g (61%), melting point: >220°C.
A solution of 6.7 g (25 mMol) of 2-n-propyl-benzimidazole-5-carboxylic acidhemisulphate and 4.9 g (25 mMol) of 2-methylaminoaniline dihydrochloride in 200 g of polyphosphoric acid is stirred for 5 h at 150°C, then poured onto 600 ml of water and made alkaline with concentrated ammonia whilst cooling with ice. The resulting solution is extracted three times with 200 ml of ethyl acetate, the crude product thus obtained is purified by column chromatography (300 g of silica gel; eluant:methylene chloride/methanol = 15:1). Yield of 2-n-propy1-5-(1-methylbenzimidazol-2-yl)-benzimidazole: 2.8 g of oil (39%).
A solution of 2.0 g (6.9 mMol) of 2-n-propyl-5-(1-methylbenzimidazol-2-yl)- benzimidazole and 0.91 g (7.5 mmol) of potassium tert-butoxide in 50 ml of dimethylsulfoxide is stirred for 90 min at room temperature, then 2.6 g (7.5 mMol) of tert-butyl 4'-bromomethyl-biphenyl-2-carboxylate are added and the mixture is stirred for a further 15 h at room temperature. The mixture is then poured onto 300 ml of water and extracted three times with 50 ml of ethyl acetate. The crude product obtained after evaporation of the organic phase is purified by column chromatography (300 g silica gel; eluant:methylene chloride/methanol = 30:1). In this way, 2.7 g (70%) of an isomer mixture are obtained (by NMR spectroscopy), contains about 1.18 g of tert-butyl-4'-[(2-npropyl- 5-(1-methylbenzimidazol-2-yl)-benzimidazol-1-yl)-methyl]biphenyl-2- carboxylate and about 1.52 g of tert-butyl 4'-[(2-n-propyl-6-(1- methylbenzimidazol-2-yl)-benzimidazol-1-yl)-methyl]biphenyl-2-carboxylate).
2.70 g of the isomer mixture obtained above are dissolved in 100 ml of methylene chloride, mixed with 40 ml of trifluoroacetic acid and stirred for 4 h at room temperature. The mixture is then evaporated to dryness in vacuo, the residue is dissolved in 100 ml of 2 N sodium hydroxide solution, the solution is washed with 50 ml of diethylether and the product mixture is precipitated by acidifying the aqueous phase with acetic acid. By column chromatography (400 g of silica gel, eluant:methylene chloride/methanol = 15:1) of the solid thus obtained 0.9 g (74%) of Telmisartan, melting point 217°-218°C.
Therapeutic Function
Antihypertensive
General Description
Telmisartan, 4'-[(1,4'-dimethyl-2'-propyl[2,6'-bi-1H-benzimidazol]-1'-yl)methyl]-[1,1'-biphenyl]-2-carboxylic acid (Micardis), does not appear to bear any structuralrelationship to this class, but there is actually a great dealof overlap in the chemical architecture with other agents. Thefirst, and most significant, difference is the replacement of theacidic tetrazole system with a simple carboxylic acid. Thisacid, like the tetrazole, plays a role in receptor binding. Thesecond difference is the lack of a carboxylic acid near the imidazolenitrogen that also contributes to receptor binding.As with irbesartan, however, there is not a need for this groupto be acidic but, rather, to be one that participates in receptorbinding. The second imidazole ring, much like a purine basein deoxyribonucleic acid (DNA), can hydrogen bond with theangiotensin II receptor.
Biochem/physiol Actions
Telmisartan is a non-peptide AT1 angiotensin receptor antagonist.
Clinical Use
Angiotensin-II antagonist:
Hypertension
Prevention of cardiovascular events
Synthesis
Telmisartan can be prepared in eight steps starting with methyl 4-amino-3-methyl benzoate; the first and second cyclization into a benzimidazole ring occur at steps 4 and 6 respectively.
Drug interactions
Potentially hazardous interactions with other drugs
Anaesthetics: enhanced hypotensive effect.
Analgesics: antagonism of hypotensive effect and increased risk of renal impairment with NSAIDs; hyperkalaemia with ketorolac and other NSAIDs.
Antihypertensives: increased risk of hyperkalaemia, hypotension and renal impairment with ACE-Is and aliskiren.
Cardiac glycosides: concentration of digoxin increased.
Ciclosporin: increased risk of hyperkalaemia and nephrotoxicity.
Diuretics: enhanced hypotensive effect; hyperkalaemia with potassium-sparing diuretics.
ESAs: increased risk of hyperkalaemia; antagonism of hypotensive effect.
Lithium: reduced excretion (possibility of enhanced lithium toxicity).
Potassium salts: increased risk of hyperkalaemia.
Tacrolimus: increased risk of hyperkalaemia and nephrotoxicity.
Metabolism
Telmisartan is metabolised by conjugation to the glucuronide of the parent compound. No pharmacological activity has been shown for the conjugate.
Telmisartan is excreted almost entirely in the faeces via bile, mainly as unchanged drug.
Clinical claims and research
In several clinical studies, Telmisartan, at a once daily dosage, produced effective and sustained blood-pressure lowering effects with a low incidence of side effects (particularly treatment-related cough associated with ACE inhibitors in elderly patients).
Telmisartan synthesis
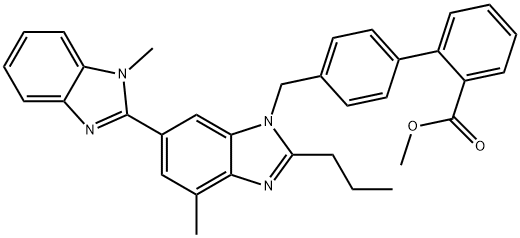
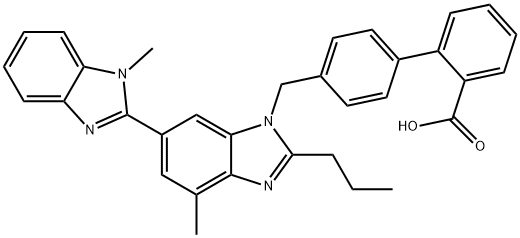
Synthesis of Telmisartan from EXECUTE STANDARD
Dosage Recommendations
The dosage of Telmisartan varies based on the condition being treated, the severity of symptoms, and the patient’s response to the medication. It is essential to follow the prescribed dosage and administration instructions provided by a healthcare professional. General dosage guidelines include:
Hypertension
Initial Dose: The typical starting dose is 40 mg taken once daily.
Maintenance Dose: Based on response, the dose may be increased to 80 mg once daily.
Maximum Dose: The maximum recommended dose is 80 mg per day.
Cardiovascular Risk Reduction
The usual dose for reducing cardiovascular risk is 80 mg taken once daily.
Heart Failure and Diabetic Nephropathy
The dosage for these conditions may vary, and it is crucial to follow the specific instructions provided by a healthcare provider.
Administration
Telmisartan tablets should be taken orally with a full glass of water. They can be taken with or without food. Taking the medication at the same time each day helps maintain consistent blood levels.
Potential Side Effects
While Telmisartan is generally well-tolerated, it can cause side effects in some individuals. Side effects may range from mild to severe.
Common Side Effects
Dizziness or Lightheadedness: Especially when first starting the medication or increasing the dose.
Fatigue: Feeling unusually tired or weak.
Nausea: Mild stomach discomfort or nausea.
Back Pain: Some patients report experiencing back pain.
Serious Side Effects
If you experience any of the following serious side effects, seek medical attention immediately:
Kidney Problems: Symptoms include changes in the amount of urine, swelling in the ankles or feet, and sudden weight gain.
High Potassium Levels: Symptoms include muscle weakness, slow or irregular heartbeat.
Severe Allergic Reactions: Symptoms include difficulty breathing, swelling of the face, lips, tongue, or throat, and severe rash or itching.
Hypotension: Symptoms include severe dizziness or fainting, particularly after starting the medication or increasing the dose.
© 2025. All rights reserved by Ross & Fleming India Super-specialty Clinics